The MiOXSYS® System
Uncovering the other side of fertility
MiOXSYS® – the most complete, definitive and rapid seminal oxidative stress measurement system
- MiOXSYS® result represents an integrated measure of all oxidants and all antioxidants in the sample, making it a clinically valuable measure.
- MiOXSYS® rapidly provides sORP (static oxidative-reductive potential) values that are predictive of male fertility potential with PPV of >95%1
- MiOXSYS® is recommended as a diagnostic system for MOSI (Male Oxidative Stress Infertility).2
- The only one such machine on the market with extensive clinical research and patent protection.

The most complete measure of Oxidative Stress
sORP (static oxidation-reduction potential) – the most complete measure of Oxidative Stress
sORP is an integrated measure of the balance between total levels of oxidants and antioxidants in a biological system or sample such as the human semen.
- It is a measure of the balance between total oxidant to total reductant activity in semen.
- It reflects the oxidative relationship between the sperm cell and its environment.
Oxidation-reduction — which is also known as a redox reaction — is a process that involves transferring electrons from a reductant to an oxidant utilizing a specific equation that has been validated within ORP software.

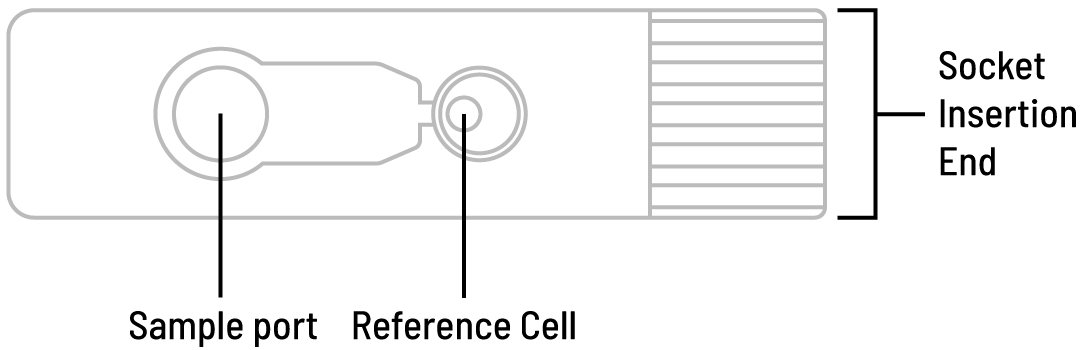
MiOXSYS® sensor
MiOXSYS® sensor: basis for the most accurate ORP measurement:
- Single use, platinum-based, stable state electrode sensor with an Ag/AgCl reference cell
- The seminal plasma is absorbed through the filter on the sample port and migrates across the working electrode to fill the reference cell, thereby completing the electrochemical circuit.
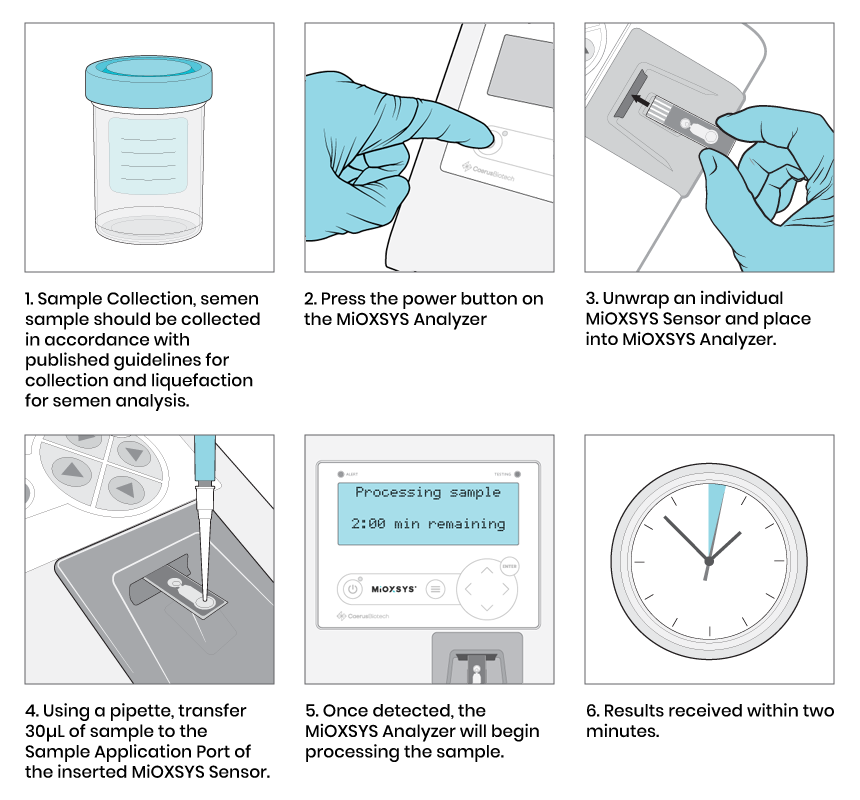
Fast and easy-to-use
MiOXSYS®: fast and easy-to-use test delivering results in as quickly as 5 minutes
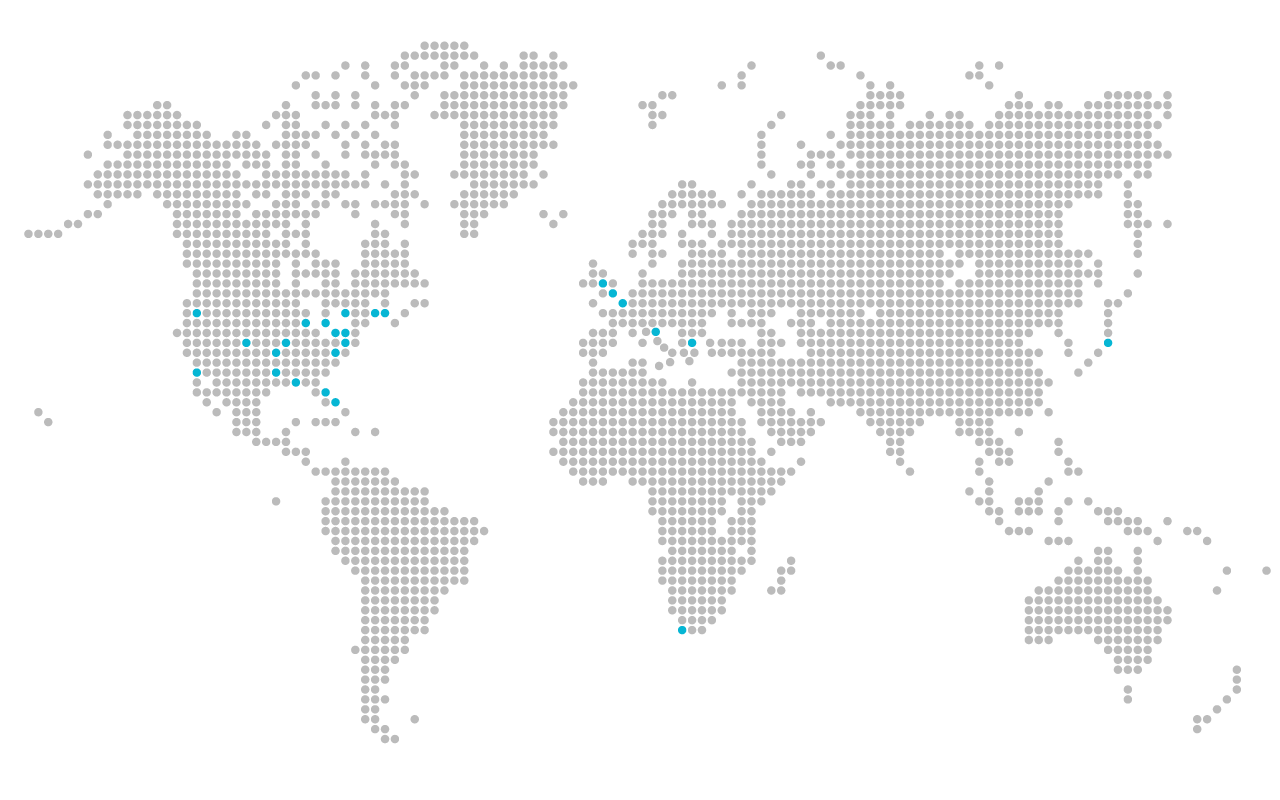
Clinical development
The analyzer system and software have been validated with over 10,000 clinical samples.
Academic institutions involved in MiOXSYS clinical development and validation, include:
Africa: University of Western Cape (South Africa).
Asia: Dokkyo Medical Center (Japan).
Europe: University of Kent (UK), University of Birmingham (UK), University of Nottingham (UK), Amien University (France), University of Bologna (Italy), University of Thessaly (Greece).
North America: Cleveland Clinic, University of Colorado, Baylor Medical Center, NYU Medical Center, University of Oregon State, Princeton University, University of Nebraska, University of Pittsburgh, University of Rochester, University of California San Diego, University of Florida, University of Kansas, University of Michigan, University of Washington, Tulane University, University of Virginia and University of Miami (in the United States); and University of Toronto (Canada).
Request a MiOXSYS® System
Want to learn more about the MiOXSYS® System?
References
1 Arafa M, Agarwal A, Al Said S, Majzoub A, Sharma R, Bjugstad KB, AlRumaihi K, Elbardisi H. Semen quality and infertility status can be identified through measures of oxidation-reduction potential. Andrologia. 2018 Mar;50(2). doi: 10.1111/and.12881. Epub 2017 Aug 3. PMID: 28771782.
2 Agarwal A, Parekh N, Panner Selvam MK, et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J Mens Health. 2019;37(3):296-312. doi:10.5534/wjmh.190055